Projects funded by Slovenian Research Agency (ARRS)
Current projects:
The kiss of death for key apoptosis players: development of BCL-2 and BAX PROTACs (J1-2485, 1.9.2020-31.8.2023)
Principal Investigator: Assist. Prof. Dr. Izidor Sosič
Link to project website
Development of innate immune receptor-targeting chimeras as custom-tailored vaccine adjuvants (J3-2517, 1.9.2020-31.8.2023)
Principal Investigator: Assoc. Prof. Dr. Žiga Jakopin
Link to project website
Photochemistry toolbox for discovery of advanced ATP-competitive chemical probes with Topoisomerase IIalpha inhibitory activity (N1-0172, 1.1.2021-31.12.2023)
Principal Investigator: Prof. Dr. Janez Ilaš
Link to project website
A Covalent Approach Against Antibiotic Resistance (N1-0169, 1.1.2021-31.12.2023)
Principal Investigator: Prof. Dr. Stanislav Gobec
Link to project website
Development of antibacterial compounds targeting validated enzymes in peptidoglycan biosynthesis (J1-2484, 1.9.2020-31.8.2023)
Principal Investigator: Prof. Dr. Stanislav Gobec
Link to project website
Modular asymmetric total synthesis of bioactive multi-chiral natural products (Z1-2635, 1.9.2020-31.8.2022)
Principal Investigator: Assist. Prof. Dr. Andrej Emanuel Cotman
Link to project website
Radiopharmaceuticals with antagonistic activity on CCK2R (J1-2483, 1.9.2020-31.8.2023)
Principal Investigator: Prof. Dr. Marko Anderluh
Link to project website
Discovery and mechanism of action of novel hEag1 potassium channel lead molecules with anti-cancer activity (N1-0098, 1.1.2019-31.12.2022)
Principal Investigator: Prof. Dr. Lucija Peterlin Mašič
Link to project website
Development of novel Hsp90 inhibitors with anticancer activity (J1-1717, 1.7.2019-30.6.2022)
Principal Investigator: Assoc. Prof. Dr. Tihomir Tomašič
Link to project website
Past projects:
Development of new carbamate-based imaging probes for cholinesterases (NC-0009, 1.4.2020-31.3.2021)
Principal Investigator: Prof. Dr. Stanislav Gobec
Link to project website
Targeted covalent inhibitors: inhibiting monoamine oxidase through non-catalytic amino-acid residues (Z1-1859, 30.6.2019-1.7.2021)
Principal Investigator: Assist. Prof. Dr. Damijan Knez
Link to project website
Identification of non-peptidic inhibitors of the immunoproteasome using fragment based drug discovery methods (N1-0068, 1.11.2017-31.10.2020)
Principal Investigator: Prof. Dr. Stanislav Gobec
Link to project website
Butyrylcholinesterase inhibitors for alleviating symptoms of Alzheimer's disease (Z1-9195, 1.7.2018-30.6.2021)
Principal Investigator: Assist. Dr. Urban Košak
Link to project website
New anticancer leads for emerging cancer target potassium ion channels hEag1 and its validation in lymphoma tumors (J1-9192, 1.7.2018-30.6.2021)
Principal Investigator: Prof. Dr. Lucija Peterlin Mašič
Link to project website
Development of NOD2 agonists and dual NOD2/TLR7 agonistic conjugates as novel vaccine adjuvants (J3-9256, 1.7.2018-30.6.2021)
Principal Investigator: Assoc. Prof. Dr. Žiga Jakopin
Link to project website
Development of multifunctional compounds for treatment of Alzheimer's disease (L1-8157, 30.4.2017-30.4.2020)
Principal Investigator: Prof. Dr. Stanislav Gobec
Link to project website
Combating bacterial resistance: optimisation of bacterial cell wall biosynthesis inhibitors (L1-6745, 1.7.2014-30.6.2017)
Principal Investigator: prof. dr. Stanislav Gobec
Infections caused by antibiotic-resistant bacteria continue to challenge health-care systems worldwide. We face growing resistance of Gram-positive and Gram-negative pathogens that cause infections in hospitals and in the community, with the so-called antibiotic-resistant ‘superbugs’ that now represent a major global health problem. It has been reported that 25,000 Europeans die each year as a direct consequence of infections with multidrug resistant strains of pathogenic bacteria, with an estimated economic impact of 1.5 billion Euro per year. However, the pipeline for new drugs is small, because the major pharmaceutical companies have largely abandoned antibiotic discovery even though new drugs are urgently needed. The obstacles of having few new antimicrobials on the horizon and facing increasing frequency of multidrug resistance mean that we must redouble our efforts in the search for new antimicrobials. It is imperative that we continue to search for new antibacterial drugs through innovative screening methods, and undertake rational drug design using the advances provided by protein crystal structures.
Peptidoglycan is a macromolecule that is essential for and specific to the bacterial cell wall. The enzymes involved in its biosynthetic pathway constitute potential targets for the discovery of new antibiotics. During the past 15 years, the group of PI Gobec has developed a number of important peptidoglycan biosynthesis inhibitors, the most important of which are inhibitors of the Mur enzymes (MurA to MurF) and DdlB, and of penicillin-binding proteins. In this Project, we will push forward the development of these inhibitors and optimise their on-target activities, and physico-chemical and pharmacokinetic properties.
Our approach to optimisation of inhibitors will combine the extensive knowledge within our team and our international collaborators of target enzymes and their inhibition, with cutting edge drug discovery and medicinal chemistry techniques. The Project will also benefit from detailed structural analyses, which will enable rapid in-silico design of improved inhibitor scaffolds, and subsequently, identification of novel and potent inhibitors with antibacterial activity. The optimisation of inhibitors is based on an iterative approach including computational design, and synthesis of libraries of inhibitor analogues, followed by biochemical, structural, microbiological, preliminary pharmacokinetic and toxicological evaluation. We expect to develop at least two optimised lead compounds or clinical candidates with potential to be taken forward in collaboration with the pharmaceutical industry, or to form the basis for creation of a Start-up Company. These are very important steps towards development of new broad-action antibacterials that will be of key importance in the fight against bacterial resistance. It is thus the intention of this Project to fill the gap between the burden of infections due to multidrug-resistant bacteria and the decreasing level of new antibiotics in development.
Selected publications:
1. Hrast Martina, Rožman Kaja, Jukič Marko, Patin Delphine, Gobec Stanislav, Sova Matej. Synthesis and structure-activity relationship study of novel quinazolinone-based inhibitors of MurA. Bioorganic & Medicinal Chemistry Letters, doi: 10.1016/j.bmcl.2017.05.064.
2. Rožman Kaja, Lešnik Samo, Brus Boris, Hrast Martina, Sova Matej, Patin Delphine, Barreteau Hélène, Konc Janez, Janežič Dušanka, Gobec Stanislav. Discovery of new MurA inhibitors using induced-fit simulation and docking. Bioorganic & Medicinal Chemistry Letters 2017, 27, 944-949; doi: 10.1016/j.bmcl.2016.12.082.
3. Šink Roman, Kotnik Miha, Zega Anamarija, Barreteau Hélène, Gobec Stanislav, Blanot Didier, Dessen Andréa, Contreras-Martel Carlos. Crystallographic study of peptidoglycan biosynthesis enzyme MurD: domain movement revisited. PloS One 2016,11, 3, doi: 10.1371/journal.pone.0152075.
4. Sosič Izidor, Anderluh Marko, Sova Matej, Gobec Martina, Mlinarič-Raščan Irena, Derouaux Adeline, Amoroso Ana, Terrak Mohammed, Breukink Eefjan, Gobec Stanislav. Structure-activity relationships of novel tryptamine-based inhibitors of bacterial transglycosylase. Journal of Medicinal Chemistry 2015, 58, 9712-9721, doi: 10.1021/acs.jmedchem.5b01482.
5. Hrast Martina, Sosič Izidor, Šink Roman, Gobec Stanislav. Inhibitors of the peptidoglycan biosynthesis enzymes MurA-F. Bioorganic chemistry 2014, 55, 2-15, doi: 10.1016/j.bioorg.2014.03.008.
Theoretical and practical Aspects of Terminology and Terminography in Connection with Internet Terminological Resources (L6-4045, 1.7.2011 - 30.06.2014)
The research project involves theoretic and applicative work: researching and developing terminological and terminographical theory (terminological definitions, individual parts-of-speech in terminology, e.g. the verb in legal terminology, antonymy, researching the influence of English on Slovenian with an emphasis on terminology, Slovenianisation of terminology), publication of results in monographs, articles, lectures, creating new and updating existing computer tools for processing terminology (SlovarRed 2.1), creating and publishing specialised corpora, processing, translating and standardising the terminologies of various fields, compiling and publishing normative explanatory mono- and multilingual dictionaries in paper and electronic form (the internet), organising Slovenian and international professional meetings, conducting terminological counceling. We also intend to cooperate with the Slovenians living abroad and important European terminological centres.
Preclinical development of new antibacterial drugs (L1-4039,1.7.2011 - 30.06.2014)
Principal Investigator: prof. dr. Stanislav Gobec
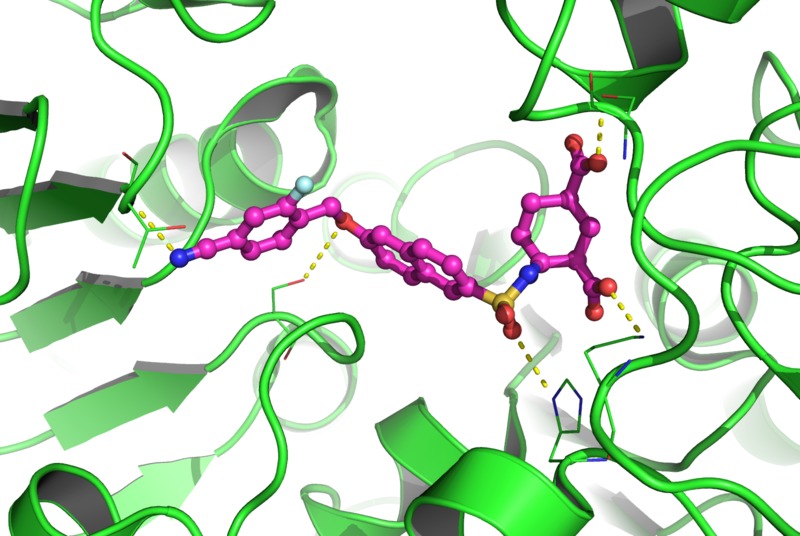
As society has become increasingly dependent upon antibiotics, the selection pressure for emergence of resistant bacterial strains has increased, and so-called antibiotic-resistant ‘superbugs’ are now a major global health problem. The development of resistance can interfere with, or even prevent, the treatment of bacterial infections, especially in hospitals. New drugs are urgently needed to provide doctors with fresh options to treat these infections. We intend to discover new inhibitors which will attack the formation of bacterial peptidoglycan, and this will either kill the bacteria directly or make them more susceptible to existing antibiotics.
Peptidoglycan is a macromolecule which is essential for and specific to the bacterial cell wall. The numerous enzymes involved in its biosynthetic pathway constitute potential targets for the discovery of new antibiotics. To identify novel lead compounds that are important for antibacterial drug discovery, we will target the selected enzymes that catalyze the intracellular stages (Mur ligases MurC-F and DdlB) and extracellular stages (the penicillin-binding proteins) of peptidoglycan biosynthesis. Strategies for inhibitor discovery will be based on the available X-ray crystal structures and will involve sophisticated computational techniques, like de novo structure-based inhibitor design and virtual high-throughput screening, and chemical synthesis of libraries of potential inhibitors and their biochemical (evaluation of inhibitory activities), structural (determination of X-ray crystal structures) and antimicrobial (determination of MIC values) evaluation. It is intended to discover several structurally distinct lead molecules with micromolar or nanomolar inhibition of target enzymes and with antimicrobial activities.
The strength of this project includes (1) the multi-targeted approach that involves enzymes from a variety of pathogenic species, and (2) the vast number of techniques employed, provided by the different types of highly complementary expertise of all partners incuded. The group of the partners from Slovenia is specialised in computational drug discovery methods like virtual high-throughput screening and de novo molecular design, medicinal chemistry and synthetic organic chemistry, while other international partner groups are specialised in bacterial peptidoglycan biosynthesis enzymes expression and purification, biochemistry, microbiology and X-ray crystallography. During the previous collaboration with international partners and Slovene pharmaceutical company Lek d.d. we have developed a series of promising inhibitors of bacterial peptidoglycan biosynthesis enzymes and this work will be countinued during this project. Lead compounds identified in this project will represent very important steps towards the development of new broad action antibacterials that will be of key importance in the fight against emerging bacterial resistance.
Selected publications:
1. Sosič Izidor, Anderluh Marko, Sova Matej, Gobec Martina, Mlinarič-Raščan Irena, Derouaux Adeline, Amoroso Ana, Terrak Mohammed, Breukink Eefjan, Gobec Stanislav. Structure-activity relationships of novel tryptamine-based inhibitors of bacterial transglycosylase. Journal of medicinal chemistry 2015, 58, 9712-9721, doi: 10.1021/acs.jmedchem.5b01482.
2. Perdih Andrej, Hrast Martina, Barreteau Hélène, Gobec Stanislav, Wolber Gerhard, Šolmajer Tomaž. Inhibitor design strategy based on an enzyme structural flexibility: a case of bacterial MurD ligase. Journal of chemical information and modeling 2014, 54, 1451-1466. doi: 10.1021/ci500104m.
3. Hrast Martina, Anderluh Marko, Knez Damijan, Randall Christopher P., Barreteau Hélène, O'neill Alex J., Blanot Didier, Gobec Stanislav. Design, synthesis and evaluation of second generation MurF inhibitors based ona cyanothiophene scaffold. European Journal of Medicinal Chemistry 2014, 73, 83-96. doi: 10.1016/j.ejmech.2013.11.031.
4. Hrast Martina, Turk Samo, Sosič Izidor, Knez Damijan, Randall Christopher P., Barreteau Hélène, Contreras-Martel Carlos, Dessen Andréa, O'Neill Alex J., Mengin-Lecreulx Dominique, Blanot Didier, Gobec Stanislav. Structure-activity relationships of new cyanothiophene inhibitors of the essential peptidoglycan biosynthesis enzyme MurF. European Journal of Medicinal Chemistry 2013, 66, 32-45. doi: 10.1016/j.ejmech.2013.05.013.
5. Hrast Martina, Vehar Blaž, Turk Samo, Konc Janez, Gobec Stanislav, Janežič Dušanka. Function of the D-Alanine:D-Alanine ligase lid loop : a molecular modeling and bioactivity study. Journal of medicinal chemistry 2012, 55, 6849-6856. doi: 10.1021/jm3006965.
Projects funded by EU
Current projects:
PhD4GlycoDrugH2020-MSCA-ITN-2017-EJD-765581 (H2020 MSCA ITN)
Duration of the project: 2017 – 2021
Coordinator: Prof. Dr. Marko Anderluh
Project website
The PhD4GlycoDrug consortium has a main goal to offer a European Joint Doctorate educational training network in Glyco-Drug Discovery and Development. The network combines 6 academic partners with long tradition in PhD training and accredited doctoral studies in Organic Chemistry, Medicinal Chemistry, Structural Biology and Pharmacology/Pharmacy. Non-academic partners, 4 SMEs and a research institute also constitute the consortium to offer intersectoral exposure and quality training in transferable skills to entrepreneurial ESRs. So far, only a handful of registered drugs originate from glycoscience area. To pursue an innovative research project in such a timely scientific field of great interest for pharmaceutical industry, a specific set of competences is needed. Our network will focus on delivering expert researchers highly attractive for employment by the European Pharma-industry.
Past projects:
INTEGRATE - Interdisciplinary Training Network for Validation of Gram-Negative Antibacterial Targets (H2020 MSCA ITN)
Duration of the project: 2015 – 2018
Slovenian PI: prof. dr.Danijel Kikelj
Antimicrobial resistance is posing a continuously-rising threat to global health. Indeed, one key recommendation from the recent “Action plan against the rising threats from Antimicrobial Resistance” report (submitted by the Commission to the European Parliament and Council (15.11.2011)) is the development of effective antimicrobials or alternatives for treatment of human and animal infections. The INTEGRATE project is a direct response to this. We have assembled a team of ten beneficiaries from eight EU member states, encompassing both academic and nonacademic sectors and different disciplines, to form a consortium committed to training Early Stage Researchers (ESRs) in the discovery and preclinical validation of novel Gram-negative antibacterial agents and antibacterial targets. The principle aim of the consortium is to provide a training platform where students are exposed to every aspect of the antimicrobial discovery process, ranging from target identification and validation, through organic synthesis, in silico design and compound screening, to mode-of-action and possible resistance mechanisms. This exposure will be accomplished through a concrete secondment plan, coupled with a series of high-level consortiumwide training events and networking programmes. Our intention is to reverse the current fragmentation of approaches towards antibacterial discovery through mutual cooperation. The INTEGRATE training framework is built on an innovative research project aimed at targeting important but non-essential gene products as an effective means of reducing bacterial fitness, thereby facilitating clearance of the pathogen by the host immune system. To achieve this, the individual work programmes have been designed to seamlessly inter-mesh contributions from the fields of in silico design, organic synthesis, molecular biology and biochemistry, and the very latest in vitro and in vivo screening technologies.
Recent publications:
1. Tomašič Tihomir, Mirt Matic, Barančokova Michaela, Ilaš Janez, Zidar Nace, Tammela Päivi, Kikelj Danijel. Design, synthesis and biological evaluation of 4,5-dibromo-N-(thiazol-2-yl)-1H-pyrrole-2-carboxamide derivatives as novel DNA gyrase inhibitors. Bioorganic & Medicinal Chemistry 2017, 25, 338-349. doi: 10.1016/j.bmc.2016.10.038.
2. Jakopin Žiga, Ilaš Janez, Barančokova Michaela, Brvar Matjaž, Tammela Päivi, Sollner Dolenc Marija, Tomašič Tihomir, Kikelj Danijel. Discovery of substituted oxadiazoles as a novel scaffold for DNA gyrase inhibitors. European Journal of Medicinal Chemistry 2017, 130, 171-184. doi: 10.1016/j.ejmech.2017.02.046.
3. Gjorgjieva Marina, Tomašič Tihomir, Barančokova Michaela, Katsamakas Sotirios, Ilaš Janez, Tammela Päivi, Peterlin Mašič Lucija, Kikelj Danijel. Discovery of benzothiazole scaffold-based DNA gyrase B inhibitors. Journal of Medicinal Chemistry 2016, 59, 8941-8954, doi: 10.1021/acs.jmedchem.6b00864.
ORCHID (EU FP7)
Duration of the project: 1.1.2011 – 31.12.2014
Slovenian PI: prof. dr. Stanislav Gobec
An ambitious four-year project to discover and develop potential new medicines against multiple drug resistance tuberculosis (TB) has been launched by an international consortium of 12 diverse groups.
The ORCHID alliance (Open Collaborative Model for Tuberculosis Lead Optimization) brings together TB expertise from academia, government research centres, non-for-profit organizations and small biotechnology companies with funding from the EU’s Seventh Framework Programme for Research (FP7). Under the leadership of the pharmaceutical company GlaxoSmithKline, more than 25 scientists worldwide will pool their expertise to investigate the potential of three different areas of research, all of which have shown potential activity against TB.
The aim is to complete drug discovery research to have identified and validated a novel candidate molecule ready to be tested in humans. It is only through the collaboration of such diverse experts group that all three areas can be progressed in a coordinated fashion.
The three areas to be investigated are:
- β-lactams – new evidence suggests a novel approach could make these broad spectrum antibioitics effective against TB. This approach will seek to design new animal models to demonstrate the potential of B-lactams as treatments against TB.
- New InhA inhibitors – GSK in collaboration with TB Alliance have recently identified a chemical series that can inhibit recognised known TB target (InhA), which is essential for the bacterium’s survival. Tool compounds in this series have shown activity against multi drug resistant strains (MDR-TB) in the lab and have also demonstrated promising activity in animal models of Mycobacterium tuberculosis.
- Whole-cell hits – Compounds that have been shown to be active against TB in high through put screening, but their mode of action is unknown. The project will aim to understand this activity.
Selected publications:
1. Rožman Kaja, Sosič Izidor, Fernandez-Menendez Raquel, Young Robert J., Mendoza-Losana Alfonso, Gobec Stanislav, Encinas Lourdes. A new 'golden age' for the antitubercular target InhA. Drug Discovery Today 2016, 1-29, doi: 10.1016/j.drudis.2016.09.009.
2. Pajk Stane, Živec Matej, Šink Roman, Sosič Izidor, Neu Margarete, Chung Chun-Wa, Martínez-Hoyos María, Pérez-Herrán Esther, Álvarez-Gómez Daniel, Álvarez-Ruíz Emilio, Mendoza-Losana Alfonso, Castro-Pichel Julia, Barros David, Ballell-Pages Lluís, Young Robert J., Convery Maire A., Encinas Lourdes, Gobec Stanislav. New direct inhibitors of InhA with antimycobacterial activity based on a tetrahydropyran scaffold. European Journal of Medicinal Chemistry 2016, 112, 252-257, doi: 10.1016/j.ejmech.2016.02.008.
3. Šink Roman, Sosič Izidor, Živec Matej, Fernandez-Menendez Raquel, Turk Samo, Pajk Stane, Alvarez-Gomez Daniel, Lopez-Roman Eva Maria, Gonzalez Cortes Carolina, Rullas-Trincado Joaquin, Angulo-Barturen Iñigo, Barros David, Ballell Pages Lluís, Young Robert J., Encinas Lourdes, Gobec Stanislav. Design, synthesis and evaluation of new thiadiazole- based direct inhibitors of enoyl acyl carrier protein reductase (InhA) for the treatment of tuberculosis. Journal of Medicinal Chemistry 2015, 58, 613-624, doi: 10.1021/jm501029r.
MAREX (EU FP7)
Duration of the project: 2010 – 2014
Slovenian PI: prof. dr.Danijel Kikelj
Biodiversity in the seas is only partly explored, although marine organisms are excellent sources for many industrial products. Through close co-operation between industrial and academic partners, the MAREX project will collect, isolate and classify marine organisms, such as micro- and macroalgae, cyanobacteria, sea anemones, tunicates and fish from the Atlantic, Pacific and Indian Oceans as well as from the Mediterranean, Baltic and Arabian Seas. Extracts and purified compounds of these organisms will be studied for several therapeutically and industrially significant biological activities, including anticancer, anti-inflammatory, antiviral and anticoagulant activities by applying a wide variety of screening tools, as well as for ion channel/receptor modulation and plant growth regulation. Chromatographic isolation of bioactive compounds will be followed by structural determination. Sustainable cultivation methods for promising organisms, and biotechnological processes for selected compounds will be developed, as well as biosensors for monitoring the target compounds. The work will entail sustainable organic synthesis of selected active compounds and new derivatives, and development of selected hits to lead compounds.
The project will expand marine compound libraries. MAREX innovations will be targeted for industrial product development in order to improve the growth and productivity of European marine biotechnology. MAREX aims at a better understanding of environmentally conscious sourcing of marine biotechnology products and increased public awareness of marine biodiversity and potential. Finally, MAREX is expected to offer novel marine-based lead compounds for European industries and strengthen their product portfolios related to pharmaceutical, nutraceutical, cosmetic, agrochemical, food processing, material and biosensor applications.
Selected publications:
1. Tomašič Tihomir, Katsamakas Sotirios, Hodnik Žiga, Ilaš Janez, Brvar Matjaž, Šolmajer Tomaž, Montalvão Sofia, Tammela Päivi, Banjanac Mihailo, Ergović Gabrijela, Anderluh Marko, Peterlin Mašič Lucija, Kikelj Danijel. Discovery of 4,5,6,7-tetrahydrobenzo[1, 2-d]thiazoles as novel DNA gyrase inhibitors targeting the ATP-binding site. Journal of Medicinal Chemistry 2015, 58, 5501-521, doi: 10.1021/acs.jmedchem.5b00489.
2. Hodnik Žiga, Peterlin Mašič Lucija, Tomašič Tihomir, Smodiš Domen, D'Amore Claudio, Fiorucci Stefano, Kikelj Danijel. Bazedoxifene scaffold-based mimetics of solomonsterols A and B as novel pregnane X receptor antagonists. Journal of Medicinal Chemistry 2014, 57, 4819-4833, doi: 10.1021/jm500351m.
3. Zidar Nace, Jakopin Žiga, Madge David J., Chan Fiona, Tytgat Jan, Peigneur Steve, Sollner Dolenc Marija, Tomašič Tihomir, Ilaš Janez, Peterlin Mašič Lucija, Kikelj Danijel. Substituted 4-phenyl-2-aminoimidazoles and 4-phenyl-4,5-dihydro-2-aminoimidazoles as voltage-gated sodium channel modulators. European Journal of Medicinal Chemistry 2014, 74, 23-30, doi: 10.1016/j.ejmech.2013.12.034.
4. Peigneur Steve, Žula Aleš, Zidar Nace, Chan-Porter Fiona, Kirby Robert, Madge David J., Ilaš Janez, Kikelj Danijel, Tytgat Jan. Action of clathrodin and analogues on voltage-gated sodium channels. Marine drugs 2014, 12, 2132-2143, doi: 10.3390/md12042132.
5. Tomašič Tihomir, Hartzoulakis Basil, Zidar Nace, Chan Fiona, Kirby Robert W., Madge David J., Peigneur Steve, Tytgat Jan, Kikelj Danijel. Ligand- and structure-based virtual screening for clathrodin-derived human voltage-gated sodium channel modulators. Journal of Chemical Information and Modeling 2013, 53, 3223-3232, doi: 10.1021/ci400505e.
INTAFAR (EU FP6)
Slovenian PI: prof. dr. Stanislav Gobec
The introduction of antibiotics in the medical practice has drastically reduced illness and death from infectious diseases. However, bacteria have exhibited a remarkable capacity to become resistant to commonly used antibacterial compounds. In parallel to efforts aimed at better understanding the strikingly diverse mechanisms they use, it is important to design strategies which will allow to counteract them and to search for new targets, ideally essential and specific to bacterial physiology.
The approach followed in EUR-INTAFAR is based on the well established facts that peptidoglycan biosynthesis and cell morphogenesis are related phenomena and that they are totally specific to bacterial cells, without even remotely equivalent processes in eukaryotic cells.
By attempting to interfere with these processes by inhibiting enzyme activities or perturbing protein-protein interactions, one should be able to design new antibacterial compounds active against pathogenic organisms such as streptococci, staphylococci, enterococci or chlamydiae.
Selected publications:
1. Sosič Izidor, Barreteau Hélene, Simčič Mihael, Šink Roman, Cesar Jožko, Zega Anamarija, Golič Grdadolnik Simona, Contreras Martel Carlos, Dessen Andréa, Amoroso Ana, Joris Bernard, Blanot Didier, Gobec Stanislav. Second-generation sulfonamide inhibitors of D-glutamic acid-adding enzyme: activity optimisation with conformationally rigid analogues of D-glutamic acid. European Journal of Medicinal Chemistry 2011, 46, 2880-2894. doi: 10.1016/j.ejmech.2011.04.011.
2. Tomašič Tihomir, Zidar Nace, Šink Roman, Kovač Andreja, Blanot Didier, Contreras-Martel Carlos, Dessen Andréa, Müller-Premru Manica, Zega Anamarija, Gobec Stanislav, Kikelj Danijel, Peterlin-Mašič Lucija. Structure-based design of a new series of D-Glutamic acid-based inhibitors of bacterial UDP-N-acetylmuramoyl-L-alanine:D-glutamate ligase (MurD). Journal of medicinal chemistry 2011, 54, 4600-4610. doi: 10.1021/jm2002525.
3. Turk Samo, Verlaine Olivier, Gerards Thomas, Živec Matej, Humljan Jan, Sosič Izidor, Amoroso Ana, Zervosen Astrid, Luxen André, Joris Bernard, Gobec Stanislav. New noncovalent inhibitors of penicillin-binding proteins from penicillin-resistant bacteria. PloS one 2011, 6, e19418. doi: 10.1371/journal.pone.0019418.
4. Kovač Andreja, Konc Janez, Vehar Blaž, Bostock Julieanne M., Chopra Ian, Janežič Dušanka, Gobec Stanislav. Discovery of new inhibitors of D-alanine:D-alanine ligase by structure-based virtual screening. Journal of medicinal chemistry 2008, 51, 7442-7448. doi: 10.1021/jm800726b.
5. Humljan Jan, Kotnik Miha, Contreras-Martel Carlos, Blanot Didier, Urleb Uroš, Dessen Andréa, Šolmajer Tomaž, Gobec Stanislav. Novel naphthalene-N-sulfonyl-D-glutamic acid derivatives as inhibitors of MurD, a key peptidoglycan biosynthesis enzyme. Journal of medicinal chemistry 2008, 51, 7486-7494.